The evolution of technology has allowed heart rate monitoring devices to become smaller and smaller, putting them within the reach of each individual whether through a cell phone, small dedicated devices or even watches that allow continuous monitoring of the rhythm and performing an electrocardiogram.
There are two main forms of monitoring, one of which is based on plethysmography, either by light and cell phone cameras, sensors attached to fingertips, or on watches. This plethysmography technique allows even non-purpose cell phones to obtain a reasonably accurate heart rate reading, allowing heart rate counting and identification of rhythm irregularities that may be associated with atrial fibrillation. However, it is important to note that whenever an arrhythmia is detected in plethysmography-based devices, it must be confirmed by electrocardiogram.
The second way, more precise, but requiring adapted or dedicated devices, consists of reading the electrocardiogram itself. The devices contain electrodes that allow the capture of electrical signals from the heart, being able to record two leads when there are only two electrodes, but being able to record up to six leads in devices with more than two electrodes. This technology associated with artificial intelligence systems allows, in addition to recording the ECG, its interpretation, being able to alert the patient of the appearance of atrial fibrillation, high or low heartbeats.
Recently, the European Heart Rhythm Association (EHRA) prepared, with the contribution of LAHRS, a document entitled “How to use digital devices to detect and manage arrhythmias: a practical guide for EHRA” (1) where the characteristics and applications of these devices are described, discussed in a very interesting and in-depth way, including recommendations based on expert consensus are presented in this document.
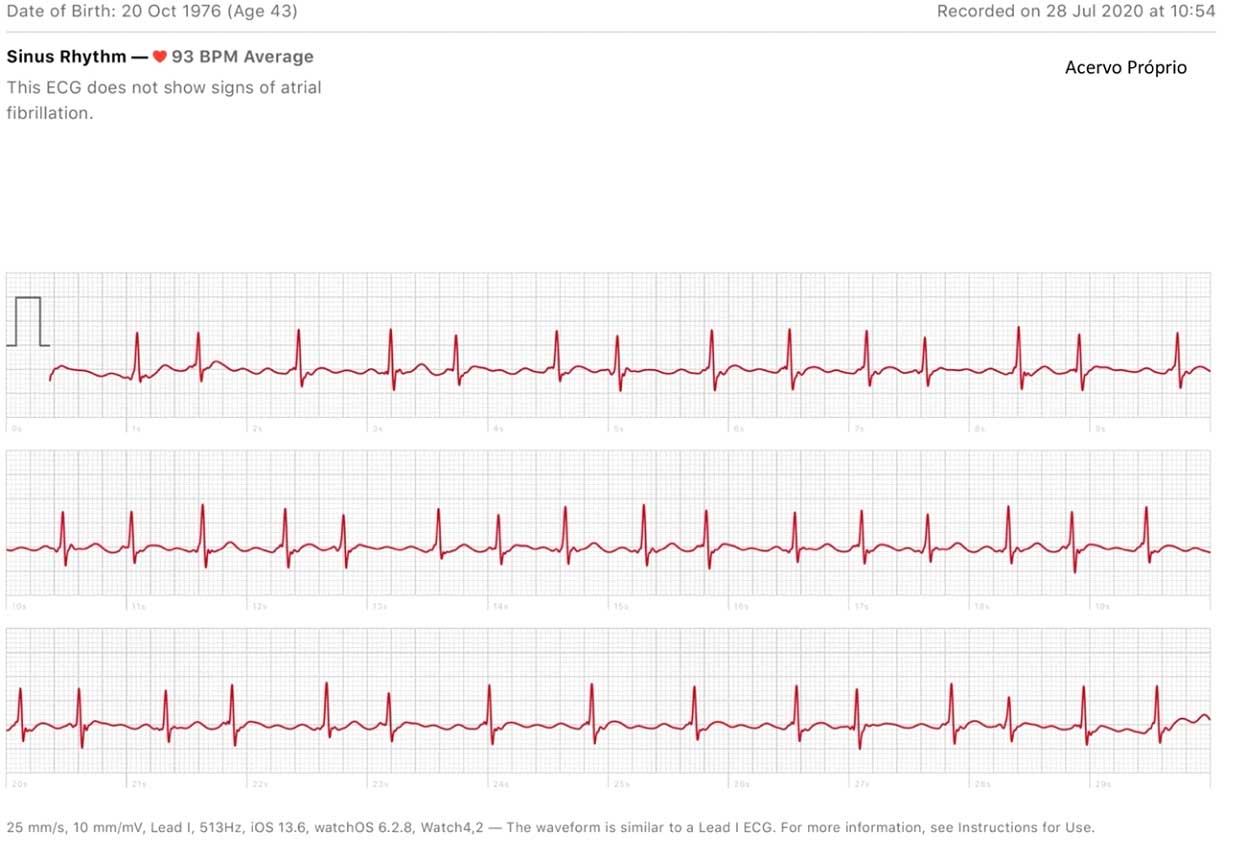
Regarding the monitoring of Atrial Fibrillation (Figure 1), the intermittent use of these devices is indicated and is beneficial in people over 75 years of age, and they can also be used in people over 65 years of age with comorbidities that increase the risk of stroke. In younger or lower-risk individuals, sporadic screening can be performed, but in these cases, due to the very low incidence of AF, they have low detection rates. An important point regarding the detection of AF is that, when it was identified and evaluated in relation to the use of anticoagulants and risk factor management, there was evidence of benefits in the prevention of embolic events. (2)
Figure 1 – Electrocardiogram demonstrating atrial fibrillation detected on a watch with electrocardiogram recording
In patients with a previous diagnosis of AF, these devices also play an interesting role in patient management. The use of these devices can contribute to improving commitment and adherence to the ABC (3) strategy for the management of AF. Another very interesting use of these devices is after the AF ablation procedure, with the aim of detecting cases of recurrence during follow-up, which allows for better drug adjustment.
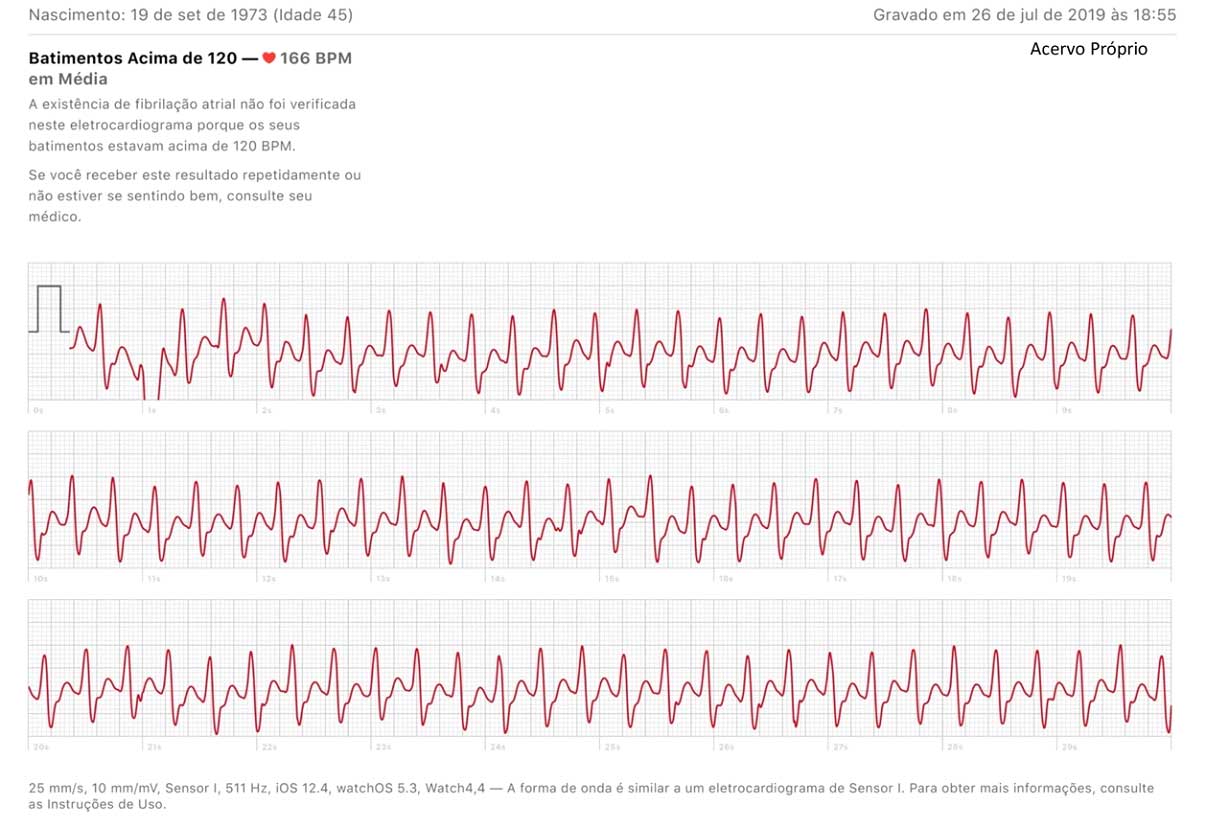
Another population where devices can help conventional treatment are patients with ventricular arrhythmias and syncope (Figure 2). However, one of the limitations of these devices is that they do not promote continuous electrocardiographic monitoring, and must be activated or inserted by the patient. In such cases, self-adhesive patch-based devices with continuous monitoring may be beneficial. Patients with paroxysmal palpitations lasting a few minutes, generally when they go to the emergency room to record the arrhythmia, they revert before arriving and, as the episodes are sporadic, often cannot be recorded on Holter or symptomatic event monitor. Currently, with the use of detection devices, it is easy to record the arrhythmia tracing, often identifying supraventricular tachycardias that are susceptible to ablation and complete control of the arrhythmia.
Figure 2 – Documented ventricular tachycardia in a patient with ischemic cardiomyopathy, inferior scarring, and episodes of short-duration rhythmic tachycardia palpitations. The recorded episode was hemodynamically well tolerated and lasted approximately 5 minutes with spontaneous reversal. The patient underwent ventricular tachycardia ablation in evolution, with no recurrence after ablation
Currently, more and more patients will carry these devices, bringing benefits in the diagnosis and treatment of cardiac arrhythmias, so it is extremely important that the doctors involved know the characteristics, advantages and disadvantages of each device, as well as its use in the different cardiac arrhythmias, being this publication of great relevance for the doctors of our society.
1. Svennberg E, Tjong F, Goette A, Akoum N, Di Biase L, Bordachar P, et al. How to use digital devices to detect and manage arrhythmias: an EHRA practical guide. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2022.
2. Svennberg E, Friberg L, Frykman V, Al-Khalili F, Engdahl J, Rosenqvist M. Clinical outcomes in systematic screening for atrial fibrillation (STROKESTOP): a multicentre, parallel group, unmasked, randomised controlled trial. Lancet. 2021;398(10310):1498-506.
3. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomstrom-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. European heart journal. 2021;42(5):373-498.